Recursion Pharmaceuticals: Machine Learning in Drug Discovery

Recursion Pharmaceuticals is deploying machine learning to deeply understand the interactions between genes, proteins, and chemicals to inform not only future drug discovery and drug repurposing, but biological life as we know it.
Machine learning is taking over modern drug discovery, and Recursion Pharmaceuticals is on that cutting edge.
Q1: By virtue of continuously feeding large volumes of data of cellular images to its already massive databases, Recursion Pharmaceuticals has “developed a massive database of biological images, each of which is relatable over time to all the others we produce” [1]. Recursion relies on the inflow of cellular data of different disease pathways and cell types, which refines its understanding of the linkages between different disease pathways. Machine learning is thus critical to the process improvement of better understanding biological targets, where it is used to model “computational changes to identify changes in cellular and sub-cellular structure in the presence of various biological perturbations” [2]. One output of this machine learning is a set of hypotheses of where certain existing drugs may be repurposed to target new reactive centers. This creates a more efficient process for developing the initial ‘funnel’ of opportunities of drug repurposing for pharmaceutical company customers interested in the additional business opportunities of its developed products. Additionally, however, modeling of disease pathways would not only inform drug discovery of new small and large molecules, which has revenue upside, but it could create cost efficiencies by determining which biological pathways to not consider for drug development, thus reducing inefficient R&D spend [3]. Pharmaceutical companies could test hypotheses with Recursion to determine whether or not to explore further research in a given therapeutic area.
Q2: In the short-term, Recursion will leverage its machine learning platform to partner with pharmaceutical companies to help with their drug repurposing and discovery needs. In the medium-term, this platform will be used to inform its own internal drug discovery and potentially personalized medicine applications. Since Recursion launched in 2013, it has developed several partnerships with pharmaceutical companies to help their drug repurposing efforts. In 2016, for example, Recursion announced an agreement with Sanofi Genzyme to identify new uses for Sanofi’s clinical stage molecules across dozens of genetic diseases [4]. Recursion also uses its platform to help its pharmaceutical company partners with their own drug discovery pipelines. In 2017, for example, Recursion announced an agreement with Takeda Pharmaceutical to provide pre-clinical candidates for Takeda’s development pipeline, using its machine learning platform, where Recursion would receive a royalty payment from any future sales resulting from those successful drugs [5].
In the medium-term, Recursion has begun to establish itself as a player in its own internal drug discovery as well as personalized medicine, which aims to tailor medical treatments to patients based on their genetic profile. In 2016, for example, Recursion participated in the a government-led initiative to leverage its machine learning platform to model the effect of various treatments at a genetic level, which would then create that link between patients’ genetic makeup and the efficacy of treatments on them [6]. In the future, Recursion will use its increasingly sophisticated machine learning platform to generate its own pipeline of drug discovery targets at a more accelerated an efficient pace than its pharmaceutical company counterparts [7].
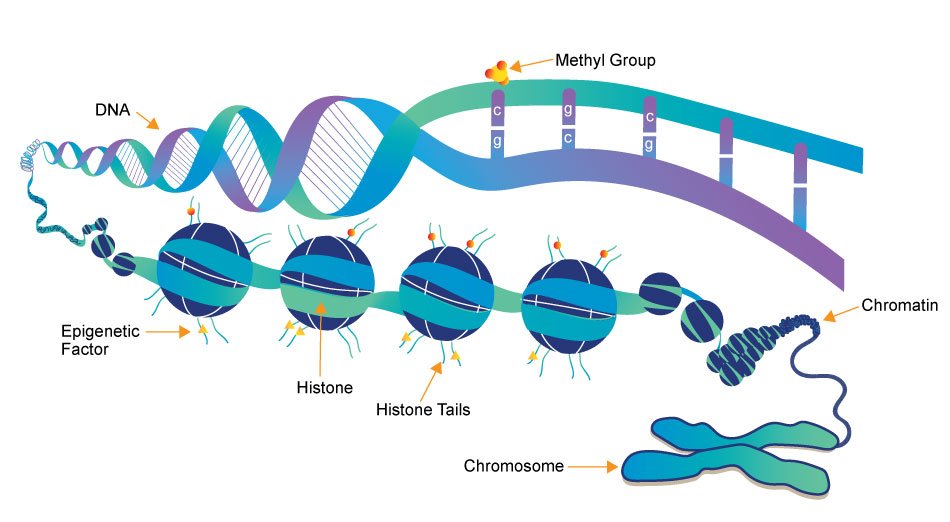
Q3: In the short-term, Recursion has an opportunity to focus on dynamically incorporating fundamentally different data, which may shift its own models of biological interactions at the cellular level. Biological research has until now focused principally on the interactions between genes, proteins, and chemicals. Recent research, however, has indicated that non-genetic modifications known as “epigenetic factors” can play a significant role in regulating gene expression and may be similarly important in modeling biological behaviors. Thus, Recursion will face the task of staying current in its understanding of biological research, which will require the incorporation of new forms of data to its machine learning platform. The medium-term consideration is similar. Recursion has an opportunity to integrate additional non-genetic factors that may play a role in disease pathways, such as dietary habits and lifestyle choices.
Q4:
(1) Given that Recursion’s computational model is only as accurate and predictive as the underlying research upon which it relies, is there an opportunity for it to use its AI to not only model existing research, but also be on the frontier of research by identifying biological breakthroughs with its algorithms?
(2) To what extent is Recursion compelled, from a policy perspective, to share these research findings, given the trade-off of: maintaining its own competitive technology advantage versus improving overall public health outcomes with socialization of research breakthroughs?
Word count: 728
[1] “Our Mission – Recursion Pharmaceuticals”. 2018. Recursion Pharmaceuticals. https://www.recursionpharma.com/our-mission/.
[2] “Recursion Pharmaceuticals Raises $60 Million To Industrialize Drug Discovery Using Artificial Intelligence”. 2018. Marketwire. http://www.marketwired.com/press-release/recursion-pharmaceuticals-raises-60-million-industrialize-drug-discovery-using-artificial-2235894.htm.
[3] Mack, Heather. 2018. “Recursion Pharma Gets Second Round This Year”. WSJ. https://www.wsj.com/articles/recursion-pharma-gets-second-round-this-year-1507030200.
[4] WIRE, BUSINESS. 2018. “Recursion Pharmaceuticals Announces Research Agreement With Sanofi Genzyme”. Businesswire.Com. https://www.businesswire.com/news/home/20160425005113/en/Recursion-Pharmaceuticals-Announces-Research-Agreement-Sanofi-Genzyme.
[5] “Recursion Pharmaceuticals Announces Research Collaboration With Takeda To Rapidly Build A Rare Disease Pipeline”. 2018. Marketwire. http://www.marketwired.com/press-release/recursion-pharmaceuticals-announces-research-collaboration-with-takeda-rapidly-build-2236525.htm.
[6] “Recursion Pharmaceuticals Supports White House Precision Medicine Initiative”. 2018. PR.Com. https://www.pr.com/press-release/659810.
[7] “Recursion Pharmaceuticals Announces $60M Series B Round”. 2018. Silicon Slopes. https://newsroom.siliconslopes.com/recursion-pharmaceuticals-announces-60m-series-b-round/.


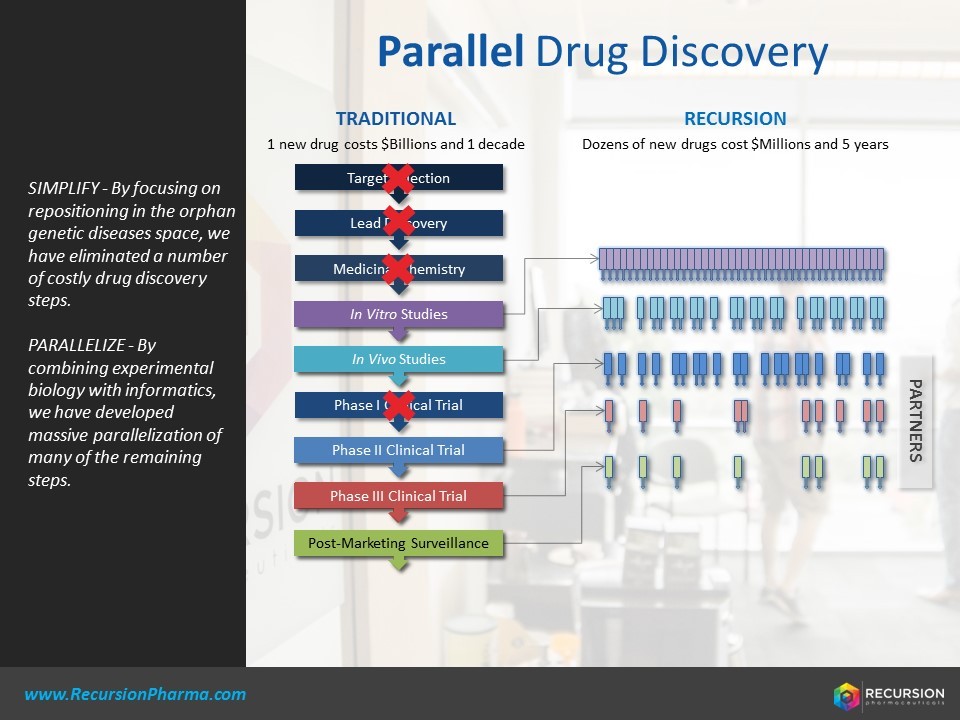


Recursion is a really interesting startup, and I applaud what they are trying to do, especially with regards to drug repurposing, which is a desperately underserved area of research and one that could bring more therapies to market faster and leaner – especially for the underserved rare and neglected diseases. One issue I see with their discovery pipeline is the use of knockdown technology to create disease models of interest, in a limited number of cell lines, rather than knockout technology in more physiologically relevant cells. The issue that arises is that it is very difficult to tell, based on their imaging technology, whether they are faithfully recapitulating the disease of interest using this technology. If the disease state isn’t faithfully generated, or it isn’t produced in a cell that is physiologically relevant to that disease (e.g. using a skin cell instead of a nerve cell), the technology may end up generating needless false positives (compounds that appear to work, but actually don’t), and a number of false negatives (drugs that could be effective, but whose activities are missed in the assays). Buttressing their initial screening results with more physiologically-relevant, stem-cell derived or patient cells may produce higher quality data and lead to more clinical candidates or repurposed drugs.
First, the pictures in this post are so helpful!
Responding to your second question, Recursion would be compelled to share these developments with FDA as they seek approval for the drugs. Outside that, they are under no legal obligation to share the technology unless portions of the basic science that underlie the technology were funded by government money (i.e. National Institutes of Health, National Science Foundation, or Department of Defense funding). In this case, the Bayh-Dole Act would allow the government to assert its ownership rights to the patent and from there they could share the technology as they saw fit.
From a social responsibility perspective, however, I think the question becomes a bit more complicated. An interesting question for the Recursion leadership team — perhaps pursuing joint ventures or partnership models where they can still reap profit but take advantage of the scale of the bigger pharmaceutical companies/spread more good would be a sufficient compromise?