A Patient’s Perspective – How Antidote is Digitalizing Clinical Trial Patient Recruitment in the US

80% of clinical trials shut down prematurely due to an inability to recruit sufficient patient numbers. Antidote is empowering patients to find relevant clinical trials, shifting the supply chain of patient enrollment.
A Patient’s Perspective – How Antidote is Digitalizing Clinical Trial Patient Recruitment in the US
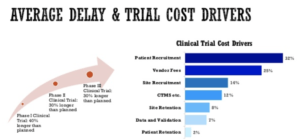
One of the primary cost drivers for pharmaceutical and biotech companies bringing drugs to market is patient recruitment and enrollment in clinical trials. Patient recruitment represents >30% of clinical trial costs [1]. In oncology specifically, 40% of all trials and 71% of phase III trials fail to recruit the targeted number of patients, delaying drug approvals and limiting patient access to trials, often during late-stage disease when patients have exhausted other care options [2].
Clinical trial patient matching historically has occurred with the use of contract resource organizations (CROs), typically private entities that pharmaceutical clients contract with to outsource management of clinical trials. By 2020, an estimated 70% of all clinical trials will be conducted by CROs. Despite the benefits of an external specialized workforce, CROs can be bureaucratic which often results in delays in drug development and rising costs [3]. Startups are emerging using digital solutions to replace a step in the CRO supply chain and more efficiently match patients to relevant trials. One tech company with a unique approach to clinical trial patient supply management is Antidote, a digital health startup with the mission to better connect patients to clinical trials.
Why Antidote’s mission?
A number of tech companies are trying to digitalize clinical trial matching, and most are approaching the problem by working with pharmaceutical companies to recruit for specific trials. For example, IBM Watson for Clinical Trial Matching uses artificial intelligence to scan patient electronic health record documents and match patient characteristics to trial inclusion criteria [4]. The supply-focused approach to clinical trial matching is important in expediting patient enrollment and facilitating drug development but it is not a solution for patients.
Antidote is filling the demand-side gap, arming patients and families looking to play an active role in care selection with information and an easy-to-use digital solution. Antidote’s primary goal is to fill the recruitment gap for trials forced to shut down prematurely due to an inability to find enough participants [5]. Antidote is streamlining information-gathering so patients can more easily search for and identify trials that match their disease specifics.
How is Antidote positioned to deliver value?
Antidote built a platform, Match, on which patients can answer simple questions about their condition and other health issues and see matching trials in their region. Antidote has partnered with 180+ patient advocacy groups (i.e., Lung Cancer Alliance, Patient Power) which embed Match on their websites so patients have easy access to the resource. Antidote developed additional tools such as Antidote Bridge which provides additional information such as financial compensation and placebo use, all information historically challenging to find due to CRO privacy [5].
In the next 2 years, Antidote is focusing on building out additional patient advocacy partnerships. Advocacy groups are strong avenues toward connecting with and educating patients but I think Antidote should also consider other direct-to-consumer marketing strategies to bolster patient awareness. Many patients do not know to ask about treatment options via clinical trials. For example, in a recent study of cancer patients, 80% of participants reported that they did not realize they had an option of clinical trials [6]. Media advertising is one of the primary ways patients learn about clinical trials, specifically for rural and inner-city patients who are historically underrepresented in clinical trials [6]. Antidote should further leverage media advertising to educate more patients about the option of clinical trials and empower them to use Match as a resource to evaluate potential experimental treatments.
In the longer-term, Antidote is working to link supply and demand of clinical trials. Information accessibility for patients is an important mission but Antidote is working on a SaaS that allows trial research sites to see which patients demonstrate potential eligibility so the research site can reach out directly to enroll those patients [7]. Additionally, I think Antidote should partner with some of the technologies like Watson to link patient enrollment supply and demand to streamline supply chain efficiencies.
Looking forward – key challenges to consider
Given the competition in this space, how can Antidote ensure its solution is complementary to supply-side models (like Watson for Clinical Trial Matching)? Pharmaceutical companies are incentivized to pump money into Watson to fill their trials as quickly as possible but how can Antidote ensure its patient-empowering niche remains?
How should companies balance patient accessibility to clinical trials in more common tumor types where there may be more patients eligible for clinical trials than can be enrolled (i.e., stage IV lung cancer or melanoma)? Right now, patient demand is limited by the fact that is challenging, costly and time consuming for patients to be enrolled in clinical trials. If trials are made more accessible, what is the role of pharma companies, regulators, and patient advocacy groups in balancing supply and demand?
(790 words)
Sources:
[1] Sen, A. (2013, September 23). “Clinical Trial Recruitment and Retention”.
[2] Krischer, J. (2017, February 28). “Experience with Direct-to-Patient Recruitment for Enrollment into a Clinical Trial in a Rare Disease: A Web-Based Study”. Retrieved November 13, 2017, from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5350442/?report=classic.
[3] Roberts, D. (2016, March 28). “Contract research organizations in oncology clinical research: Challenges and opportunities”. Retrieved November 13, 2017, from http://onlinelibrary.wiley.com/doi/10.1002/cncr.29994/full.
[4] Roberts, L. (2011, April 11). “How Watson for Clinical Trial Matching is Accelerating the Screening Process”. Retrieved November 13, 2017, from https://www.ibm.com/blogs/think/2017/04/watson-health-screening/.
[5] Baum, S. (2017, September 8). “Three approaches to enhancing clinical trial recruitment”. Retrieved November 13, 2017, from https://medcitynews.com/2017/09/three-approaches-enhancing-clinical-trial-recruitment/?rf=1.
[6] Geana, M. (2016, June 21). “Searching for cures: Inner-city and rural patients’ awareness and perceptions of cancer clinical trials”. Retrieved November 13, 2017, from http://www.sciencedirect.com/science/article/pii/S2451865416300503.
[7] Liu, A. (2017, September 13). “Clinical trial matching platform raises $11M backed by Merck’s tech fund”. Retrieved November 13, 2017, from https://www.fiercebiotech.com/cro/clinical-trial-matching-platform-raises-11m-backed-by-merck-s-tech-fund.



How does Antidote make money, and do they bare any risk of making recruited patients aware of all the risks of signing up for a clinical trial?
Laura, I definitely agree with your suggestion that Antidote should have a partnership approach in order to solve the convoluted task of matching patients with clinical trials. More recently several companies such have pushed to incorporate new technologies such as Artificial Intelligence (AI) to address the matching process of clinical trials s (https://goo.gl/4BqT2J) by mining a large amount of data.
From your description, I can see that Antidote has made a great contribution to the pharmaceutical industry, patients, and science in general by maintaining those clinical trials that would not have been possible if CRO’s had not found optimal patients to continue them. The fact that this startup company has developed extensive and strong ties with key organization as the ones you mentioned is an outstanding milestone because it breaks down the information silos and bridges the gap between two parts of the equation that are equally necessary to each other: high-quality patients and relevant clinical trials.
In fact, digitizing such a large amount of critical health-related data is probably one of the most daunting tasks, especially when it is collected directly from patients or their relatives, and that is where I have a problem with this approach. Until now I have been fairly agnostic about the quality of medical information collected directly from patients, as they usually lack a sense of the big picture of the company and are easily manipulated by the type of entries or questions posed by the platform (e.g. website or app). However, I think Antidote is a powerful source by connecting two populations that have been historically apart despite needing each other at the core. Possibly, Antidote could retrieve medical records directly from hospitals across the nation or even all over the world (with prior patient consent), and sum forces with well-developed algorithms such as Watson to generate highly relevant results that can be further filtered to offer both parties (patients and CRO’s) extremely precise patient information for specialized clinical trials, reducing the large expenditure in this exhausting matching process.
I see a bright future in the optimization of this supply chain given the imperative necessity of saving lives while advancing science and paired with an important financial muscle from the pharmaceutical industry. Several large healthcare companies such as Roivant Sciences had devoted important resources to create new companies in this space (https://goo.gl/yx2hT1) and I cannot wait to see how these efforts on digitalization in the medical field unfolds!
One push-back I had when reading the part about direct-to-customer is when we think of things like WebMD, where people can now look up their symptoms and they’re given a list of potential diseases. While this is useful in terms of both patient and doctor awareness-building, I do think that this will need to be used with doctor supervision to prevent potential issues (or clinical trials being overwhelmed by people who think they’re sick).
To answer your first question, Antidote has an inherent edge over Watson in that you already have willing patients for your trial. With Watson, the pharmaceutical companies would have to pay for Watson to find potential patients, then convince those patients to join the trial. With Antidote, the self-selected list of patients is there for the trial to pick. The cost savings are significant, and may be worth the slight delay.
On your second question, I don’t believe that is Antidote’s role to pre-screen patients for an over-demanded trial. Antidote is simply a match making service, and isn’t responsible for the patient after relaying the name to the research group.
I think Antidote is an interesting idea, but I’m concerned about getting the initial patient pool along with their medical records. Medical record storage is already complicated, and people are hesitant to post their information on non-professional looking sites. I would advise Antidote to start marketing hard.
This is a very interesting company that is helping address a much needed problem. I’m excited to see what they are doing. However, I’m very curious how patient confidentiality and acts like HIPAA that restrict access to patient data play a role in this scenario. I’m interested to hear how IBM Watson’s Clinical Trial Matching is getting around these limitations and if there is some way that patients are opting into their data being included in these searches. I also know there have been issues in the past with marketing directly to patients in health care and I’m curious how this works here.
Hey Laura,
Thank you for bringing this company to our attention, clinical trials are very important. If you are interested in clinical trails and how technology can help facilitate them – you should check out some of the work that Apple is doing. It’s not a perfect system, but a lot of the things that Apple is trying with ResearchKit are pretty cool. As for Antidote – I definitely agree with you that they need to do more direct to consumer advertising. I could see this company really have an impact if they can reach more patients. As for your second question, I’m not exactly concerned about it at the moment. Clinical Trials are so supply-deficient that I don’t see it being a problem very soon.