BlueStar – Managing diabetes one click at a time
For diabetes type 2 patients, the app “Bluestar” has been a revolutionary way to manage daily life, making it a great example of the transformative impact of mobile health.
Fitbit, 23andMe, Pillpack – digital business models and products are undeniably breaking into every dimension of healthcare. Funding for digital health has risen dramatically over the past few years, jumping from $1.1 billion to $4.5 billion between 2011 and 2015. (1) Bluestar – dubbed “one of the 5 Healthcare Tech Startups to Watch in 2016” by Forbes – is a tangible example of how digital technologies change patient’s lives and help them achieve better outcomes at a lower cost. (2)
Differentiated business model and operating model
Diabetes presents a real and increasing problem in the developed world. Today in the US, 25.8 million people or 8.3% of the population are living with the disease, resulting in an estimated cost of $245 billion, including $176 billion in direct medical costs and $69 billion in reduced productivity. (3)
Until recently type 2 patients had to determine when to test their blood glucose levels and take tablets or doses of insulin largely by themselves. Bluestar presents the first mobile prescription therapy approved by the FDA and reimbursed by health insurers – a true revolution in the healthcare space. Prescribed by physicians just like normal drugs, the app plays on the various aspects of mobile technology to simplify patient’s management of the disease. (4)(5)
The app offers virtual “real-time, motivational, behavioral and educational coaching”. (6) Patients are asked to regularly input their blood glucose levels, the amount of carbs consumed, doses of insulin injected and their mood. Building off the patient’s historic data and Bluestar’s proprietary analytic systems, the patient then receives immediate, personalized guidance, like for example the suggestion to eat 15g carbohydrates, paired with options of what portions of food would fulfill the nutritional requirement. As such, Bluestar acts as a reliable, judgment-free advisor in the patient’s daily actions and helping the patient so that the disease does not have to be constantly on top of his mind.
Since all results are stored and analyzed, patients are able to track their progress over time and motivated to take additional steps to follow a healthy lifestyle. A wide array of available information on nutrition and exercising supports these endeavors.
Bluestar also facilitates entirely new ways of communicating with clinical stakeholders. In the app, patients can text-message with Bluestar’s remote expert team to clarify questions and concerns remotely. When patients come to see their physician, the app creates automatic reports about the patients’ “track-record” and even recommends treatment adjustments to their medication regimen if required, based on evidence from the app. Finally, patients are also reminded upcoming appointments or when it would be time to see a certain healthcare provider again.
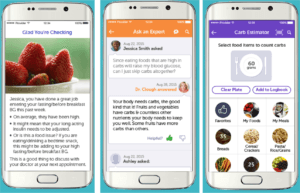
In clinical trials the app demonstrated superior outcomes compared to patients receiving usual care. Average blood sugar levels dropped by 1.9% on average for participants over the course of three months, whereas they showed only a decline of 0.7% pursuing traditional disease management. (7) Admission to emergency rooms and hospitals fell by 58% for patients using the app for more than 12 months. (8)
At the same Bluestar is priced at 30-50% less than branded diabetes drugs, which cost around $200 to $300 a month. (9) Part of those cost savings can be attributed to the fact that Bluestar had a much quicker and cheaper development cycle than traditional pharmaceutical drug companies would typically undergo. To illustrate this, clinical trials for a neurology drug evolve around $36’000 per patient, whereas digital health-start-ups spend typically only $1’000 to $5’000 per patient in a clinical trial, given that much of the extensive efforts required to recruit, manage and test patients can be done remotely. (10) Moreover, the fact that the app can be simply downloaded practically cuts out all manufacturing and distribution costs.
Additional areas for improvement
Bluestar has recently announced a partnership with J&J to link their app to LifeScan’s Bluetooth-enabled OneTouch Verio Flex blood glucose monitoring system to simplify the entry of data. (11) While this presents a great opportunity, there are further steps to transform Bluestar into a truly integrated and holistic management system for diabetes patients:
- Build-in sharing option: Giving patients the option to share their data with family and friends might serve as additional nudge to comply with their regimen
- Create a strong patient community: The Israelian app HelpAround for example connects local diabetes patients so that they can communicate and support each other as well as to identify the most pressing issues of the diabetes community (7)
- Integrate food delivery and gym apps: Bluestar could directly suggest menus from delivery services or work-outs from fitness apps with their personalized coaching
- Serve medication management platform: Notify the patient once their running low on drugs and trigger a prescription-filling
Bluestar is only one example to showcase how digital technologies our society to leapfrog to better standards of care and it remains interesting to see what innovations will follow over the next decade.
(800 words)
Sources:
(1) The Boston Consulting Group (2016): Medtech Companies Need to Transform While Times Are Still Good, https://www.bcgperspectives.com/content/articles/medical-devices-technology-transformation-medtech-companies-need-transform-while-times-still-good/
(2) Sheynin, D. (2016): 5 Healthcare Tech Startups To Watch In 2016, Forbes Brand Voice, http://www.forbes.com/sites/centurylink/2016/02/09/5-healthcare-tech-startups-to-watch-in-2016/#6675e90b5443
(3) Business Wire (2013), WellDoc Launches BlueStar, First FDA-Cleared, Mobile Prescription Therapy for Type 2 Diabetes with Insurance Reimbursement, http://www.businesswire.com/news/home/20130613005377/en/WellDoc-Launches-BlueStar-FDA-Cleared-Mobile-Prescription-Therapy
(4) The Boston Consulting Group (2014), Fulfilling the Promise of mHealth Through Business Model Innovation, https://www.bcgperspectives.com/Images/Fulfilling_the_Promise_of_mHealth_Through_Business_Model_Innovation_Apr_2014_tcm80-157211.pdf
(5) See (3)
(6) See (3)
(7) Moukheiber, Zina (2013), Trailblazer WellDoc To Sell First Mobile Prescription Therapy, http://www.forbes.com/sites/zinamoukheiber/2013/06/14/trailblazer-welldoc-to-sell-first-mobile-prescription-therapy/#74b815336b69
(8) Parmar, Arundhati (2016), 8 FDA-blessed Wearables and Apps changing healthcare, http://www.mddionline.com/article/8-fda-blessed-wearables-apps-changing-healthcare-bluestar
(9) See (7)
(10) Steinberg, D.; Horwitz, G. & Zohar, D. (2015), Building a business model in digital medicine, Nature Biotechnology, 33, 910-920, http://www.nature.com/nbt/journal/v33/n9/full/nbt.3339.html
(11) BioPharma Dive: WellDoc, LifeScan join forces on digital diabetes partnership, http://www.biopharmadive.com/news/welldoc-lifescan-join-forces-on-digital-diabetes-partnership/426286/



Thank you for sharing your insights on this company! It is very impressive that their app has proved to be so effective and has thus been FDA approved! One thing I was wondering is whether connecting patients to each other patients is actually something patients would appreciate. There are two cases to consider here: the waiting room and the online forum. I have never heard patients talk to each other about their condition in a doctor’s waiting room even though likely other patients have similar conditions when they are waiting for the same doctor to treat them. Nevertheless, there are obviously forums online where people share these things anonymously. However because this app is not at all anonymous (it requires to share a lot of personal information), I believe the case is more comparable to the waiting room situation and I am therefore not sure patients would appreciate sharing with other patients. In addition, my impression of forums where people do share such information is not necessarily very trustworthy. I would therefore not advise this company to open up for communication between users.
Google has created a prototype contact lens to measure the level of glucose in from your tear-duct. [1] We are a long way off from having this be cheap enough to compete with the OneTouch Verio Flex product you mention. But in due course someone may have invented embedded chips in our bodies to be able to input the data automatically… and at this point, the software could be bundled with the hardware to make a properly integrated product. Although, with the Theranos débâcle maybe this is too hopeful. [2]
[1] http://www.visionaware.org/blog/visionaware-blog/googles-prototype-smart-contact-lens-measuring-blood-glucose-levels-for-people-with-diabetes-1418/12
[2] http://www.wsj.com/articles/theranos-retreats-from-blood-tests-1475713848