Bioprinting – The Future of the Healthcare Industry
Organovo and other biotech companies are changing the shape of medical research and practice by developing and perfecting bioprinting technology. With the ability to print 3D tissues and organs, what are some of the opportunities and challenges presented by this technology?
Additive manufacturing, otherwise known as three-dimensional (3D) printing, has been around for almost 40 years[1], but has historically been too expensive to be adopted for widespread use. However, 3D printing has reached an inflection point, as lower costs and technological advancements have made it applicable in countless industries. Today, healthcare, aerospace & defense, and consumer goods industries drive the increasing demand for 3D printing.
Within the healthcare industry, one of the use cases of additive manufacturing is in the form of bioprinting. The most common method of bioprinting works similar to how an inkjet printer works; printers lay down layers of ink (in this case, bio-ink or biocompatible materials, cells and supporting components) to develop 3D functional living tissues[2]. Currently, the main applications of bioprinting are for drug discovery and development, toxicology testing and regenerative medicine[3].
Organovo, based in San Diego, CA, is a pioneer in bioprinting. The company specializes in bioprinting human liver tissues and kidney tissues to be used for acute and chronic toxicity studies for preclinical drug discovery and development programs. Using a proprietary 3D printing process, Organovo is able to develop multi-cellular, dynamic, and functional 3D human tissue models that remain viable for extended periods of time in vitro and exhibit key functional features that mimic aspects of natural 3D tissue. By being able to test the effects of preclinical drugs on live tissue, drug development can be accelerated as soon as the drug is shown to not be toxic to humans.
| Disruption of cell adhesion | Necrosis | Steatosis |
 |
 |
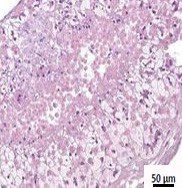 |
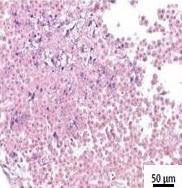
Figure 1: Analogous to human tissue biopsies, different patterns of damage can be detected histologically in ExVive™ 3D Bioprinted Liver Tissue using various treatments (H&E)[4].
In the short term, Organovo is actively partnering with leading pharmaceutical companies, like Merck[5], to allow for a more efficient (in terms of both time and money) drug development process. Pharmaceutical development is extremely costly, with average R&D costs per drug of $2.6 billion[6]. Additionally, an overwhelming majority of drugs (94%[7]) end up failing in the clinical trial phase. One of the key focusses of the FDA in drug development is around liver toxicity, which also happens to be the most common cause for discontinuing clinical trials and for withdrawal of approved drugs during the post-market surveillance stage[8]. As such, Organovo’s liver tissue product is a tool that can vastly increase the efficiency of pharmaceutical R&D spend by identifying early on whether a drug will be discontinued due intolerable to liver toxicity.
In the longer term, the company is pursuing its goal of applying its bioprinting technology to more than just preclinical drug development. Organovo sees a future where engineered tissues are used as a routine source of therapy for patients with damaged or diseased tissue. Today, they are working together with partners to do more research on the science that will allow them to make larger replacement tissues on demand[9], reducing wait times for patients looking for donors and, ultimately, curing diseases and saving lives.
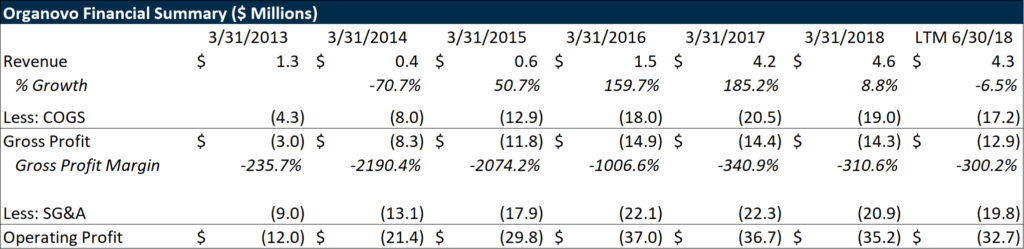
While Organovo’s business is fascinating and will benefit the overall well-being of humanity, it is currently extremely unprofitable. Bioprinting has become more economical, but there is still a long way to go to reduce costs before it can bring this business model to a self-sustainable state. Organovo continuously relies heavily on issuing equity to keep the lights on and fund their operations, having raised almost $200 million in the public markets since the company went public in 2012.
To address the near-term problem of Organovo’s products being unprofitable on a contribution margin basis, the company should work with producers of bio-ink to figure out ways to reduce production costs.
Another way that Organovo is addressing the near-term profitability issue is by joining the Advanced Regenerative Manufacturing Institute (ARMI), which recently received $300 million in public-private investment from leading manufacturers, universities, nonprofits and the federal government to develop scalable manufacturing processes for engineered tissues and organs[11]. The company can work with the other 100 partner organizations in ARMI to make the technology even more accessible.
Organovo and its peers will continue to refine bioprinting technology so that they can create more sophisticated biological structures. There are clear benefits in accelerating drug development and reducing patient wait time for transplants. However, are there any potential risks to look out for? Should this technology be regulated and by what kind of regulatory body?
[1] Arun, K. (2018). Global Markets for 3D Printing. [online] Available at: https://www-bccresearch-com.ezp-prod1.hul.harvard.edu/market-research/instrumentation-and-sensors/global-markets-for-3d-printing-ias102c.html [Accessed 9 Nov. 2018].
[2] Murphy, S. and Atala, A. (2014). 3D bioprinting of tissues and organs. Nature Biotechnology, 32(8), pp.773-785.
[3] Bergin, J. (2016). Bioprinting: Technologies and Global Markets. [online] Available at: https://www-bccresearch-com.ezp-prod1.hul.harvard.edu/market-research/biotechnology/bioprinting-markets-technologies-report-bio148a.html [Accessed 9 Nov. 2018].
[4] Organovo. (2018). Organovo – Bioprinting functional human tissue. [online] Available at: https://organovo.com/ [Accessed 9 Nov. 2018].
[5] Organovo. (2018). Merck ‘bio-inks’ deal to use Organovo’s 3D printed liver for preclinical studies – Organovo. [online] Available at: https://organovo.com/merck-bio-inks-deal-to-use-organovos-3d-printed-liver-for-preclinical-studies/ [Accessed 9 Nov. 2018].
[6] DiMasi, J., Grabowski, H. and Hansen, R. (2016). Innovation in the pharmaceutical industry: New estimates of R&D costs. Journal of Health Economics, 47, pp.20-33.
[7] Fda.gov. (2018). Step 3: Clinical Research. [online] Available at: https://www.fda.gov/ForPatients/Approvals/Drugs/ucm405622.htm [Accessed 9 Nov. 2018].
[8] Fda.gov. (2018). Liver Toxicity Knowledge Base (LTKB). [online] Available at: https://www.fda.gov/ScienceResearch/BioinformaticsTools/LiverToxicityKnowledgeBase/ucm2024036.htm [Accessed 9 Nov. 2018].
[9] Organovo. (2018). 3D Human Tissues for Medical Research & Therapeutics – Organovo. [online] Available at: https://organovo.com/tissues-services/3d-human-tissues-medical-research-therapeutics/ [Accessed 9 Nov. 2018].
[10] CapitalIQ
[11] Organovo, I. (2018). Organovo and Samsara Sciences Partner with New Manufacturing USA Institute. [online] GlobeNewswire News Room. Available at: https://globenewswire.com/news-release/2018/05/03/1495905/0/en/Organovo-and-Samsara-Sciences-Partner-with-New-Manufacturing-USA-Institute.html [Accessed 9 Nov. 2018].



Great write-up! I think the bioprinting of organs is extremely important for the reasons you’ve mentioned, but while reading your piece I also recalled a horror movie in which (if I remember correctly) the DNA of a murdered supermodel was fed into a bioprinting schematic and instead of reproducing her enviable body, the process created a monstrosity. While this scenario is obviously a gross exaggeration, it begs larger moral questions, specifically regarding the bioprinting of organs like the human brain and heart. Would a human brain, absent a body, be sentient? Could a lone human heart posess a soul? We have to discuss and regular these questions before mad scientists (joke) run amok.
Great job Michael! I believe your essay touches upon a critical topic that will dramatically change what human beings think about life. I totally agree on the enormous benefits that tissue and organ printing may entail, not only for drug testing but also for curing deadly diseases such as liver cancer.
Another relevant use of live cells printing is in food. Modern Meadow, for instance is a startup investing in printing live cells to produce meat. In 2013 the company produced its first burger that costed over $300,000.
However, I think that the free development of this technology might imply serious risks and should be consciously debated among the scientific and political leaders of the world. Let’s image the hypothetical situation that companies such as Organovo manage to profitably produce 3D printed organs, what is the next step? Create brains, hearts, bones? Print a human being? We should be aware of the powerful implications that this technology might imply for our lives both in the positive and not so positive side.
I found your piece very interesting, and fully agree that bioprinting is the future of modern health care. As you mention in the article bioprinting can be used in a number of applications, one of which is regenerative medicine. Due to an aging population and lifestyle choices adding to the burden of illness, the demand for tissue and organ transplants is increasing fast, however, due to all the advancements in medicine and car safety the supply of suitable donors is decreasing. In the US only one third of patients on the national transplant waiting list received an organ last year, leading to the death of 20 waiting individuals according to the US Government Organ Donation Statistics[1]. By successfully implementing bioprinting in clinical applications, the lives of thousands of patients could potentially be saved.
However, your article also raises a number of critical questions related to the risks of bioprinting. First of all, there is a question about the cost of potential treatments utilizing bioprinting technology. A high price could deny some patients access, and further increase patient disparity in health care. Additionally, there are concerns regarding the safety of bioprinting in clinical applications. I firmly believe that we need regulatory standards to ensure that the procedure is safe and has no bi-effects, given the limited knowledge of the effect of bioprinted tissues and organs in the human body.
In the end, the benefits of improving health care with bioprinting outweighs the risks, however, the risks can be mitigated by regulatory standards and policies governing the use of the technology.
[1] U.S. Government Information on Organ Donation and Transplantation, “Organ Donation Statistics”, https://www.organdonor.gov/statistics-stories/statistics.html, accessed November 2018
I loved reading this! Thanks for sharing. Coming from a biology background, I feel bioprinting still has a ways to go, especially given the point you raised at the end about regulations. The process of creating a human organ is even more scientifically complex than developing biologics (drug derived from a living organism). Biologics are a class of it’s own and face in-depth regulatory scrutiny due to the fact that not only must the end product or compound be scrutinized but also the process by which the biologic is created. 3-D printed organs will likely also require multiple layers of regulation, as the safety and efficacy test is not as simple as analyzing a compound and measuring dosage.
Hi Michael, thanks for sharing this interesting information! Bio-printing certainly seems like a very interesting technology, and it sounds like it will save millions in R&D cost, accelerate drug development, and reduce the need to test drugs on human subjects. It sounds like the technology still has a long way to go until it can produce full, functioning organs. This could really be a great benefit to the world by increasing the availability of organ transplants and prevent unnecessary deaths from a lack of available organs. However, I think it’s possible to take this technology too far in the future, once we have the ability to print entire organs and organ systems, could you print human beings or do things like brain transplants. There are a number of ethical questions, which we will have to work together as a society to create the frameworks for dealing with